CMR Surgical robot arm in world-first paediatric clinical trial
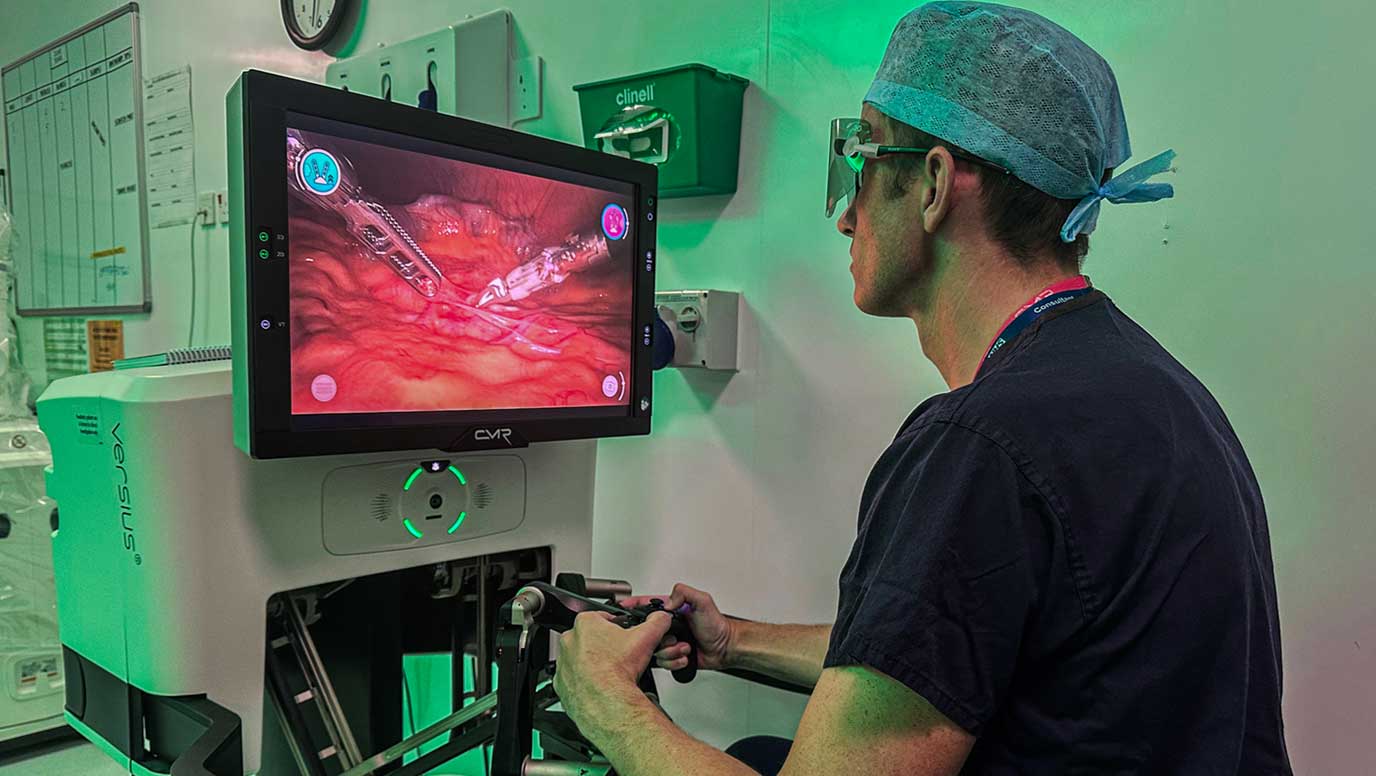
Led by Ewan Brownlee, Consultant Paediatric Urologist at the Southampton Children’s Hospital, part of the University Hospital Southampton NHS Foundation Trust, the prospective clinical study will assess the safety and efficacy of Versius for use in urological procedures in paediatric surgery.
The trial will take place over three NHS clinical sites in the UK: Southampton Children’s Hospital, Guy’s and St Thomas’ NHS Foundation Trust and Manchester University NHS Foundation Trust.
The trial focuses on children and infants under the age of 18, undergoing a range of urological procedures with Versius including but not limited to robotic assisted pyeloplasty, ureteroureterostomy, nephrectomy and Mitrofanoff formation.
The Versius Surgical Robotic System is a small, modular and versatile surgical robot used for robotic assisted minimal access surgery – a form of keyhole surgery.
The uniquely small and modular design of Versius allows the system to be set up in a way that gives the surgeon optimal access in small, hard to reach spaces within an operating zone, while maintaining clear access to the patient for the anaesthetist and wider surgical team. The design of Versius provides the opportunity to support surgeons to conduct robotic-assisted paediatric surgery, and bring the benefits to more patients.
Mark Slack, Co-founder and Chief Medical Officer at CMR Surgical saId: “The small, modular design of Versius and small instrument diameter provides an opportunity to be able to perform robotic-assisted surgery for the treatment of paediatric patients.
“We are committed to the responsible introduction of Versius across new specialties and we look forward to working with our partners as we undertake this clinical trial to ensure the safety and efficacy of Versius in paediatrics.”

Ewan Brownlee, Chief Investigator of the prospective clinical trial and Consultant Paediatric Surgeon at Southampton Children’s Hospital added: “It is a privilege to be part of such a fantastic team here in Southampton; there is a huge list of people who have helped get us to this point of being able to start the first clinical trial using Versius in children and I am truly grateful to them all.
“It has also been a pleasure to be able to collaborate with excellent colleagues in Manchester and the Evelina and with CMR to be able to deliver this trial. We are excited for what feels like a landmark step forward in the development of paediatric robotic surgery.”
This study has been evaluated and approved by the West of Scotland Research Ethics Committee, the Medicines and Health Care Products Regulatory Agency and the Health Research Authorities.
A total of 150 patients will participate in the trial which will include a thorough clinical follow up of their surgical outcomes, recovery and clinical outcomes for up to a year following surgery.
Versius has now been used in over 23,000 surgical cases, with over 130 different surgical procedures undertaken across seven surgical specialties. The system is in routine use across urology, general surgery, gynaecology, colorectal and thoracic surgery.
Versius is also being used in an ongoing clinical trial assessing the suitability of the system for use in transoral robotic surgery (TORS) at Liverpool University Hospitals NHS Foundation Trust’s (LUHFT) Aintree University Hospital.